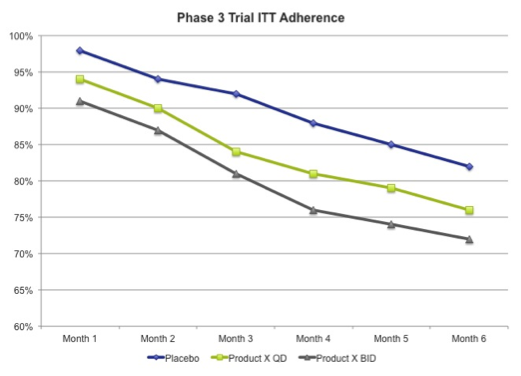
Of all the predictive questions one faces in launching a new biologic or pharmaceutical product, the ceiling of your product’s adherence curve may be one of the easiest to tackle with a high degree of accuracy. The answer to this question is buried within your clinical study report(s).
The fact is that the ceiling for your adherence curve is revealed by carefully examining the ITT (intent to treat) analysis by time increment, preferably monthly for most brands. Once you’ve plotted this curve, you can safely assure your Senior Management that it won’t get any better after launch.
But wait, why can’t your adherence curve get any better?
- HCP (i.e., $) and patient (i.e., free quality medical care) financial incentives for trial adherence are not repeatable in the real world
- Interventions (e.g., personalized patient follow up by the practice) employed are generally not scalable for use with the general disease population
- Patient population careful culled to maximize trial completion does not reflect your real world patient pool
While it may not be possible to recreate the adherence curve from the Pivotal trial(s) in the real world, an in-depth understanding of the adherence data, challenges and adherence-enhancing interventions from the Pivotal trial(s) is your first step in building a validated adherence framework for your brand.
What next? Pick up the phone and introduce yourself to the clinical study coordinator for the site that had the best percent completion for your Pivotal. I’m confident you might learn a few things!